|
R&D Systems
dgal 30 Dgal 30, supplied by R&D Systems, used in various techniques. Bioz Stars score: 93/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/dgal 30/product/R&D Systems Average 93 stars, based on 1 article reviews
dgal 30 - by Bioz Stars,
2026-03
93/100 stars
|
Buy from Supplier |
|
Bio-Techne corporation
goat anti gal3  Goat Anti Gal3, supplied by Bio-Techne corporation, used in various techniques. Bioz Stars score: 99/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/goat anti gal3/product/Bio-Techne corporation Average 99 stars, based on 1 article reviews
goat anti gal3 - by Bioz Stars,
2026-03
99/100 stars
|
Buy from Supplier |
|
Proteintech
immunosorbent assay elisa kit  Immunosorbent Assay Elisa Kit, supplied by Proteintech, used in various techniques. Bioz Stars score: 92/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/immunosorbent assay elisa kit/product/Proteintech Average 92 stars, based on 1 article reviews
immunosorbent assay elisa kit - by Bioz Stars,
2026-03
92/100 stars
|
Buy from Supplier |
|
R&D Systems
anti goat gal3 antibody  Anti Goat Gal3 Antibody, supplied by R&D Systems, used in various techniques. Bioz Stars score: 94/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/anti goat gal3 antibody/product/R&D Systems Average 94 stars, based on 1 article reviews
anti goat gal3 antibody - by Bioz Stars,
2026-03
94/100 stars
|
Buy from Supplier |
|
R&D Systems
biotinylated antibody baf1154  Biotinylated Antibody Baf1154, supplied by R&D Systems, used in various techniques. Bioz Stars score: 94/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/biotinylated antibody baf1154/product/R&D Systems Average 94 stars, based on 1 article reviews
biotinylated antibody baf1154 - by Bioz Stars,
2026-03
94/100 stars
|
Buy from Supplier |
|
R&D Systems
human gal 3 quantikine kit  Human Gal 3 Quantikine Kit, supplied by R&D Systems, used in various techniques. Bioz Stars score: 99/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/human gal 3 quantikine kit/product/R&D Systems Average 99 stars, based on 1 article reviews
human gal 3 quantikine kit - by Bioz Stars,
2026-03
99/100 stars
|
Buy from Supplier |
|
R&D Systems
recombinant human galectin 3 protein  Recombinant Human Galectin 3 Protein, supplied by R&D Systems, used in various techniques. Bioz Stars score: 94/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/recombinant human galectin 3 protein/product/R&D Systems Average 94 stars, based on 1 article reviews
recombinant human galectin 3 protein - by Bioz Stars,
2026-03
94/100 stars
|
Buy from Supplier |
|
R&D Systems
antigalectin 3  Antigalectin 3, supplied by R&D Systems, used in various techniques. Bioz Stars score: 93/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/antigalectin 3/product/R&D Systems Average 93 stars, based on 1 article reviews
antigalectin 3 - by Bioz Stars,
2026-03
93/100 stars
|
Buy from Supplier |
|
R&D Systems
anti psg1 antibodies  Anti Psg1 Antibodies, supplied by R&D Systems, used in various techniques. Bioz Stars score: 93/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/anti psg1 antibodies/product/R&D Systems Average 93 stars, based on 1 article reviews
anti psg1 antibodies - by Bioz Stars,
2026-03
93/100 stars
|
Buy from Supplier |
|
OriGene
rc208785l4v  Rc208785l4v, supplied by OriGene, used in various techniques. Bioz Stars score: 93/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/rc208785l4v/product/OriGene Average 93 stars, based on 1 article reviews
rc208785l4v - by Bioz Stars,
2026-03
93/100 stars
|
Buy from Supplier |
|
Shanghai Korain Biotech Co Ltd
elisa kit  Elisa Kit, supplied by Shanghai Korain Biotech Co Ltd, used in various techniques. Bioz Stars score: 92/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/elisa kit/product/Shanghai Korain Biotech Co Ltd Average 92 stars, based on 1 article reviews
elisa kit - by Bioz Stars,
2026-03
92/100 stars
|
Buy from Supplier |
|
Bio-Rad
rabbit anti human gal 1 igg  Rabbit Anti Human Gal 1 Igg, supplied by Bio-Rad, used in various techniques. Bioz Stars score: 90/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/rabbit anti human gal 1 igg/product/Bio-Rad Average 90 stars, based on 1 article reviews
rabbit anti human gal 1 igg - by Bioz Stars,
2026-03
90/100 stars
|
Buy from Supplier |
Image Search Results

Journal: Acta Neuropathologica
Article Title: Galectin-3 shapes toxic alpha-synuclein strains in Parkinson’s disease
doi: 10.1007/s00401-023-02585-x
Figure Lengend Snippet: Antibodies used for immunostainings
Article Snippet:
Techniques:

Journal: Acta Neuropathologica
Article Title: Galectin-3 shapes toxic alpha-synuclein strains in Parkinson’s disease
doi: 10.1007/s00401-023-02585-x
Figure Lengend Snippet: Antibodies used for western blot
Article Snippet:
Techniques: Western Blot

Journal: Acta Neuropathologica
Article Title: Galectin-3 shapes toxic alpha-synuclein strains in Parkinson’s disease
doi: 10.1007/s00401-023-02585-x
Figure Lengend Snippet: Galectin-3 (GAL3) is associated with Lewy Bodies (LB) and Pale Bodies (PB) in PD patients. a Immunofluorescence analysis of GAL3 in association with distinct forms of human α-synuclein (hSYN) aggregation. Β-sheet structure marker Methoxy-X04 was used to discriminate between LB and PB. Multiple core LB and PB are shown. GAL3 is present in both types of aggregates independently of neuromelanin presence. Scale bar 10 µm. b GAL3 is present in a diverse subset of hSYN accumulations with a precise negative correlation (blue arrows). Scale bar 10 µm. c Proportion of hSYN aggregates that are associated with GAL3. Methoxy-X04 was used as a specific marker of LB. Single (sLB) and multiple core LB (mLB) were discriminated ( p < 0.05). d Protein levels of GAL3 measured by ELISA in the Cortex of Control and PD Patients (PD-Cx) ( p < 0.001), and in the Substantia nigra (PD-SN) of PD patients
Article Snippet:
Techniques: Immunofluorescence, Marker, Enzyme-linked Immunosorbent Assay

Journal: Acta Neuropathologica
Article Title: Galectin-3 shapes toxic alpha-synuclein strains in Parkinson’s disease
doi: 10.1007/s00401-023-02585-x
Figure Lengend Snippet: GAL3 variably associates with lysosomes in the outer layers LB in all the studied patients. a GAL3 surrounding LB was found in all 6 patients studied (P.1–6). Variable amount of GAL3 vesicles was found. Note lower hSYN staining in the presence of GAL3. Scale bar 10 µm. b High resolution microscopy showed a ring-like pattern for GAL3 without any hSYN inside. Scale bar 10 µm. c Immunofluorescence analysis revealed that GAL3 is associated with recruited lysosomes (LAMP1) in the vicinities of LB. Scale bar 10 µm. d Combination of GAL3 immunohistochemistry with immunofluorescence showed that GAL3 is associated with autofluorescent lipofuscin vesicles in PD patients. Scale bar 10 µm. e Immunofluorescence analysis revealed that GAL3 accumulates inside MAP2 + neurons in the viccinities of LB. Scale bar 10 µm
Article Snippet:
Techniques: Staining, Microscopy, Immunofluorescence, Immunohistochemistry

Journal: Acta Neuropathologica
Article Title: Galectin-3 shapes toxic alpha-synuclein strains in Parkinson’s disease
doi: 10.1007/s00401-023-02585-x
Figure Lengend Snippet: Recombinant galectin-3 (Gal3) impairs synuclein aggregation in vitro. a Thioflavin-T (ThT) aggregation assay showed a rapid aggregation for recombinant human α-synuclein (αSyn) that was impaired in the presence of recombinant Gal3 (purple line). Notably, carbohydrate recognition domain (CRD) mutation (Gal3 R186S ) reverted this effect. b Proteinase K (PK) digestion at increasing concentration of resultant conditions from a) showed a lower stability in the presence of Gal3 (red line). c When Gal3 was added to αSyn pre-formed fibrils (PFF) after aggregation was completed, an increased signal was observed in the presence of ThT after 15 h. d PK digestion at increasing concentration of resultant fibrils from ( c ) showed similar stability of PFF in the presence of Gal3. e Electron microscopy images after uranyl negative staining of PFF after 24 h incubation with Gal3 (right panels). Note a marked disorganization of the fibrils network after Gal3 incubation with increased shortened species (upper right panel), and the change of morphology (lower right panel) with rounded structures attached to the fibrils. Scale bar 1 µm (upper panels) and 200 nm (lower panels). f Native PAGE Western Blot of the final results obtained in c ) Note that Gal3 promoted an increase in smaller soluble species released by αSyn fibrils. g Direct interaction of Gal3 with different αSyn species was investigated by ELISA. 2 µM Gal3 concentration were precoated in a 96 well plate and 2 µM αSyn species were incubated. 450 nm absorbance was measured to detect bounded protein. All types of species presented high affinity for Gal3 coated well compared with the control condition in absence of αSyn ( p < 0.001). No relevant absorbance was detected in the absence of precoated Gal3 (data not shown). h Addition of sonicated PFF pre-incubated with gal3 (PFFgal3) for 30 min to dopaminergic cell line N27 for 48 h led to a decreased number of cells compared with PFF alone (** p < 0.01; *** p < 0.001). i Graphical abstract representing the hypothesis proposed based on our in vitro studies about Gal3-αSyn interaction. Gal3 could impact αSyn elongation in de novo formation of fibrils while also affecting structured fibrils with little impact on the dense core but release of small species
Article Snippet:
Techniques: Recombinant, In Vitro, Mutagenesis, Concentration Assay, Electron Microscopy, Negative Staining, Incubation, Clear Native PAGE, Western Blot, Enzyme-linked Immunosorbent Assay, Sonication

Journal: Acta Neuropathologica
Article Title: Galectin-3 shapes toxic alpha-synuclein strains in Parkinson’s disease
doi: 10.1007/s00401-023-02585-x
Figure Lengend Snippet: GAL3 early overexpression leads to chronic activation and neuronal internalization. a Western Blot against GAL3 from brain homogenates from WT and Gal3KO mice revealed constitutive expression of GAL3 in WT mice. b Western Blot quantification of total GAL3 protein in WT mesencephalon samples. No difference was found between contralateral (Right hemisphere, RH) and ipsilateral (Left hemisphere, LH) hemispheres. Data are expressed as percentage fold to actin. c Double immunofluorescence 6 months after adenovirus injection showed clusters of CD11B + microglial cells highly reactive for GAL3. Internalized pSYN led to overexpression of GAL3 in WT microglia. pSYN was internalized by microglia independently of GAL3 genotype. Scale bar 20 µm. d TNFα quantification in SN and STR was performed on a MesoScale Discovery platform analysing brain extracts from AAV5-hSYN injected SN and STR ( p < 0.05). e Neuronal primary cell culture from WT mice showed efficient Gal3 internalization after incubation with 0.8 µM gal3 for 10 days. Note no difference in endogenous αSyn staining after addition of Gal3. f hSYN/GAL3 double immunofluorescence from injection area of mice WT brains 2 weeks after injection revealed no colocalization and significant upregulation of GAL3. Scale bar 50 µm. GAL3 lo /hSYN colocalization (white arrow) can be found near highly reactive GAL3 + cell indicating GAL3 release and neuronal GAL3 internalization. Scale bar 10 µm. g hSYN/GAL3 double immunofluorescence of mice WT brains 4 weeks after adenovirus injection revealed neuronal GAL3 staining. Scale bar 10 µm
Article Snippet:
Techniques: Over Expression, Activation Assay, Western Blot, Expressing, Immunofluorescence, Injection, Cell Culture, Incubation, Staining

Journal: bioRxiv
Article Title: Defining the mechanism of galectin-3-mediated TGF-β1 activation and its role in lung fibrosis
doi: 10.1101/2023.10.11.561855
Figure Lengend Snippet: Representative western blot of pSmad2 levels in (A) non-IPF HLFs (N=3) and (B) iHBECs (N=2) pre-treated with S0 μM SB-431S42 (ALKS inhibitor) or 1 μM GB0139 (galectin-3 inhibitor) for 20 minutes prior to 2-hour treatment with 10 μg/mL galectin-3 or 2 ng/mL TGF-β1. Western blot bands were quantified using densitometry analysis and presented as a ratio of pSmad2/tSmad2.
Article Snippet: After serum starvation, cells were stimulated with either 10 μg/mL
Techniques: Western Blot

Journal: bioRxiv
Article Title: Defining the mechanism of galectin-3-mediated TGF-β1 activation and its role in lung fibrosis
doi: 10.1101/2023.10.11.561855
Figure Lengend Snippet: Representative western blots of pSmad2 levels in non-IPF HLFs pre-treated with (A) NOTT199SS β1 inhibitor (0.1-100 nM) or (B-D) galectin-3 inhibitors GB0139, GB1107 and GB1211 (1 μM) or GB0149 (0.1-10 μM) for 20 minutes prior to stimulation with 2 ng/mL TGF-β1 (2-hour) or 50 μM LPA (4-hour). Cells pre-treated with S0 μM SB-431542 (ALK5 inhibitor) were included as a control demonstrating maximal inhibition of pSmad2 signaling. Western blot bands were quantified using densitometry analysis and presented as a ratio of pSmad2/tSmad2.
Article Snippet: After serum starvation, cells were stimulated with either 10 μg/mL
Techniques: Western Blot, Inhibition

Journal: bioRxiv
Article Title: Defining the mechanism of galectin-3-mediated TGF-β1 activation and its role in lung fibrosis
doi: 10.1101/2023.10.11.561855
Figure Lengend Snippet: Soluble galectin-3 (sequential injections, 19.5 - 5000 nM) binding to glycosylated or deglycosylated αv integrins: (A) αvβ1, (B) αvβS and (C) αvβ6 immobilised on the surface of a Series S sensor chip CM5 (approximately 1000 RU). (D) Soluble galectin-3 (sequential injections, 156.3-20000 nM) binding to glycosylated or deglycosylated TGFβRII immobilised to a Series S sensor chip CMS (approximately 400 RU). SPR signals were measured in RU and all sensorgrams baseline-corrected. Binding response values plotted in GraphPad Prism with connecting line/curve shown.
Article Snippet: After serum starvation, cells were stimulated with either 10 μg/mL
Techniques: Binding Assay
Journal: bioRxiv
Article Title: Defining the mechanism of galectin-3-mediated TGF-β1 activation and its role in lung fibrosis
doi: 10.1101/2023.10.11.561855
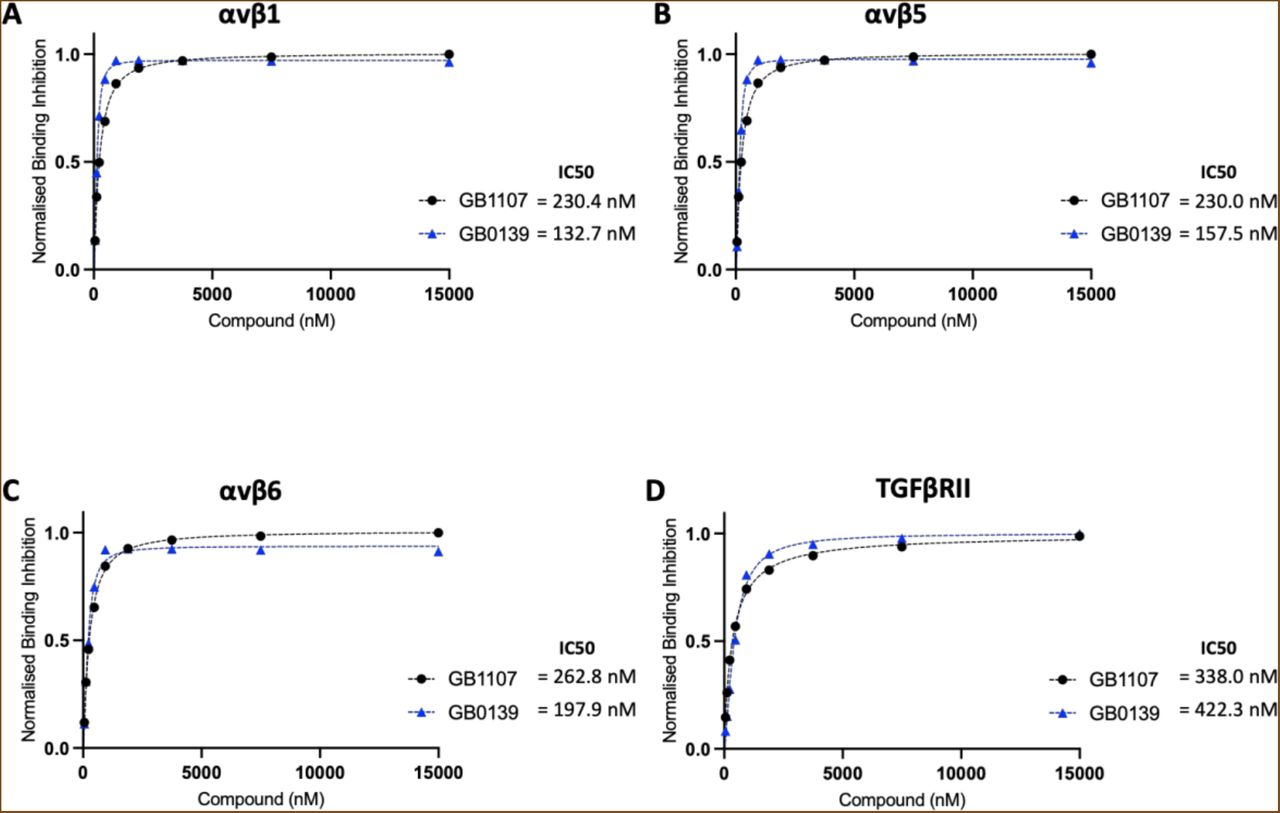
Figure Lengend Snippet: Solution competition binding assays performed with the galectin-3 inhibitor GB0139 (blue) or GB1107 (black) for αv integrins: (A) αvβ1, (B) αvβS and (C) αvβ6 or (D) TGFβRII in the presence of galectin-3 at 625 nM. Response values are normalised with respect to the highest binding response (DMSO control) and competitive inhibition graphs plotted in GraphPad Prism. IC50 values were calculated by non-linear regression analysis (binding saturation) - specific binding with hill slope.
Article Snippet: After serum starvation, cells were stimulated with either 10 μg/mL
Techniques: Binding Assay, Inhibition

Journal: bioRxiv
Article Title: Defining the mechanism of galectin-3-mediated TGF-β1 activation and its role in lung fibrosis
doi: 10.1101/2023.10.11.561855
Figure Lengend Snippet: Representative western blots showing co-immunoprecipitation of galectin-3 and the β1 integrin. Whole-cell protein lysates (6S0 μg/ IP reaction) from untreated non-IPF HLFs p6 (N=3) were immunoprecipitated with an anti-β1 integrin antibody (10 μg/ IP reaction) and immunoblotted for galectin-3 (upper panel) or immunoprecipitated with an anti-galectin-3 antibody (10 μg/ IP reaction) and immunoblotted for the β1 integrin (lower panel). Co-IP input, FT and wash steps loaded as controls. Proteins separated by reducing SDS-PAGE and target protein size estimated from the marker migration pattern.
Article Snippet: After serum starvation, cells were stimulated with either 10 μg/mL
Techniques: Western Blot, Immunoprecipitation, Co-Immunoprecipitation Assay, SDS Page, Marker, Migration

Journal: bioRxiv
Article Title: Defining the mechanism of galectin-3-mediated TGF-β1 activation and its role in lung fibrosis
doi: 10.1101/2023.10.11.561855
Figure Lengend Snippet: Representative confocal microscopy images (63x magnification) showing PLA of galectin-3 and the β1 integrin in (A) non-IPF HLFs p3-4 (N=3) or (B) IPF HLFs p3 (N=4) in the absence or presence of TGF-β1 stimulation (2 ng/mL TGF-β1 for 24 hours). Cells probed with a mouse anti-β1 integrin primary antibody (S μg/mL) and a rabbit anti-galectin-3 primary antibody (S μg/mL) followed by anti-rabbit PLUS and anti-mouse MINUS probes. Colocalization of galectin-3 and the β1 integrin 40 nm indicated by red fluorescence with DAPI counterstaining (blue).
Article Snippet: After serum starvation, cells were stimulated with either 10 μg/mL
Techniques: Confocal Microscopy, Fluorescence

Journal: bioRxiv
Article Title: Defining the mechanism of galectin-3-mediated TGF-β1 activation and its role in lung fibrosis
doi: 10.1101/2023.10.11.561855
Figure Lengend Snippet: (A) Downstream signaling of TGF-β1 following its integrin-mediated activation requires the integrin and TGF-β1 receptor to be in close proximity on the cell surface. (B) The galectin-3 carbohydrate binding domain binds to the glycosylation sites on αv integrins and the TGF-β1 receptor forming a galectin lattice at the cell surface which facilitates receptor clustering. This scaffold ensures that TGF-β1 can act on its receptor and potentiates TGF-β1 signaling. GB0139 binds to the galectin-3 carbohydrate recognition domain and blocks these protein-glycan interactions.
Article Snippet: After serum starvation, cells were stimulated with either 10 μg/mL
Techniques: Activation Assay, Binding Assay